Similarly if the knob is touch with a positively charged rod electrons will be drawn up the metal rod leaving positively charged leaves. 
since both leaves now contain negative charges they will repel. When a negatively charge object such as a negatively charged glass rod touches the knob the electric charge travels down the tube to the metal leaves. In an uncharged electroscope the leaves hang straight down. The rod is inserted into a flask with a rubber stopper (see image below). A typical classroom electroscope consists of a metal knob at top and a metal rod with thin metal leaves at the bottom. It was the first electrical measuring instrument. What is an Electroscope and What is it Used For?Īn electroscope is a scientific instrument that is used to detect the presence and magnitude of electric charge on a body. As above the lines are drawn toward the negative charge. Note that for positive charge the field lines point outward and for negative field they point inward.Įlectric Field lines between a positive and negative charge are shown below. The electric field gets stronger as we approach the particle.Įlectric fields are visualized by drawing extended lines as show in the image below. When an electron moves into the electric field of a proton it is pulled to the proton. The concept of an electric field is used to explain why charged particles exert forces on each other. The simple rule for electric charges is: Like chages repel each other and unlike charges attract each other. Similar charges will repel so negative electrons repel each other. 
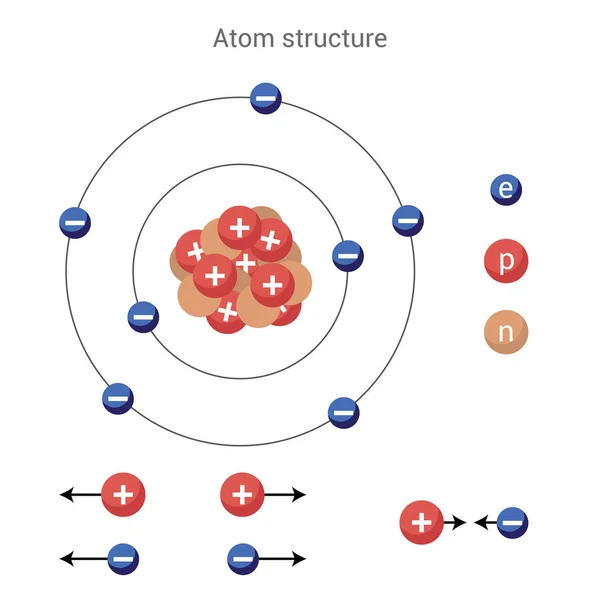
Force of attraction exists between oppositely charged particles - electrons and protons are therefore attracted to each other. A force that pushes apart particles is a repulsive force. So electrons are attracted to protons and this is what holds atoms together. A force that pulls particles together is called an attractive force. Charged particles exhibit similar behaviors of push and full. The protons are positively charged, the electrons are negatively charged, and the neutrons are neutral.The terms positive charge and negative charge were originally described by Benjamin Franklin.Ī force is defined as a push or pull. The amount or magnitude of charge on protons is the same as electrons. Charge however can be measured and it affects the behavior of particles. This property is not like other physical properties of matter. It does not contain a neutron.īoth protons and electrons have a basic property called charge. Remember that the hydrogen atom is only composed of a a single proton and electron. 
Protons and neutrons are found in the nucleus of an atom and eletrons are found in an area outside often described as an electron cloud (see what are atomic orbitals for more advanced description). In order to be neutral, an atom must have the same number of electrons and protons.We know that all objects are made up of atoms and atoms are composed of smaller particles called protons, electrons and neutrons. If a neutral atom has 10 protons, it must have 10 electrons. If a neutral atom has 2 protons, it must have 2 electrons. If a neutral atom has 1 proton, it must have 1 electron. In other words, a neutral atom must have exactly one electron for every proton. This means that the negative charge on an electron perfectly balances the positive charge on the proton. Negative and positive charges of equal magnitude cancel each other out. \)) are useful, because, as you can see, the mass of a proton and the mass of a neutron are almost exactly \(1\) in this unit system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
